Wednesday, April 22, 2015
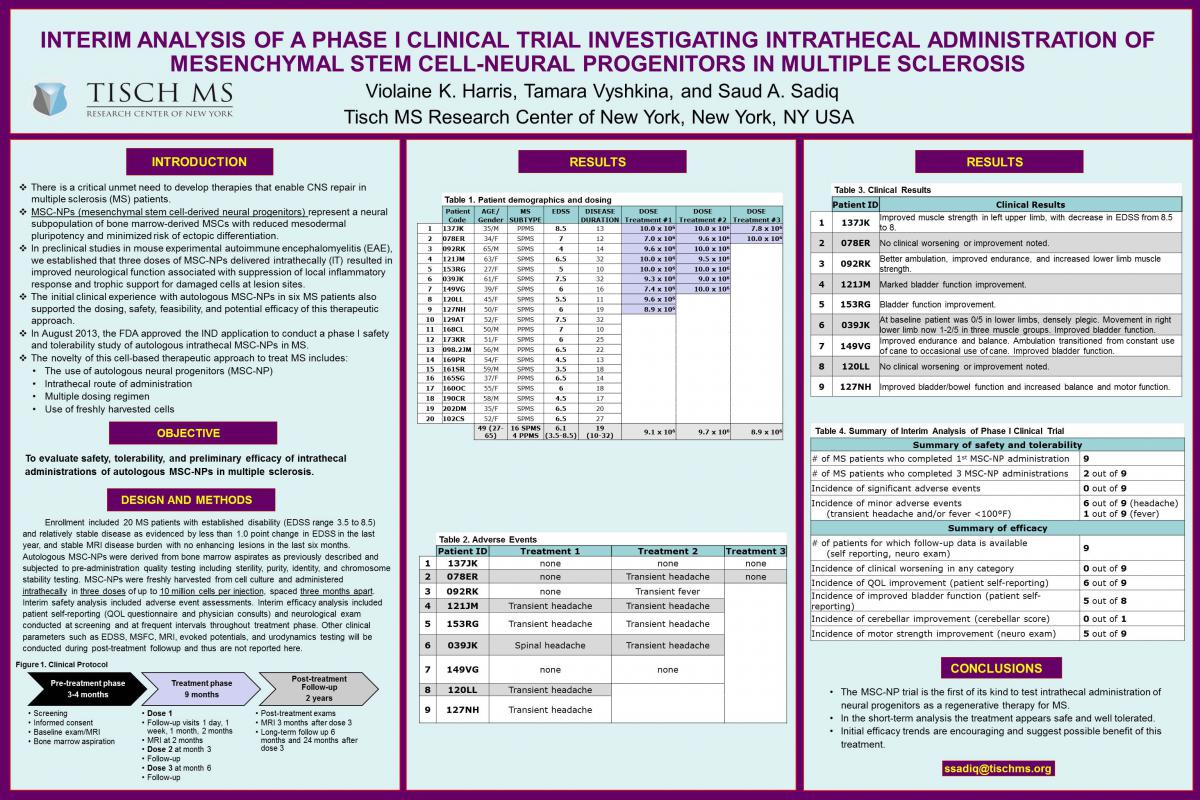
Interim Results Indicate Positive Efficacy Trends in FDA-Approved Phase I Clinical Stem Cell Trial For Patients with Multiple Sclerosis
New York, NY - April 23, 2015 - Tisch MS Research Center of New York (Tisch MSRCNY) today announced encouraging preliminary results from its FDA-approved Phase I trial using autologous neural stem cells in the treatment of multiple sclerosis (MS). MS is a chronic autoimmune disease of the central nervous system where the myelin sheath is attacked and neurodegeneration may occur. It affects more than 2.3 million people worldwide. The significant results were selected for presentation during the Multiple Sclerosis Highlights in the Field session at the 67th American Academy of Neurology (AAN) Annual Meeting in Washington, D.C.
Tisch MSRCNY research showed that in the interim analysis, six of nine patients are exhibiting increased motor strength, improved bladder function and an enhanced quality of life. Importantly, the treatments are well tolerated and thus far no serious adverse events were reported.
“This preliminary data is encouraging because in addition to helping establish safety and tolerability, the trial is yielding some positive therapeutic results even at this early stage,” said Dr. Saud A. Sadiq, Chief Research Scientist at Tisch MSRCNY and the study’s principal investigator. He cautioned however, that these results are an interim analysis and definitive conclusions will only be made upon completion of the trial.
The study investigates a pioneering regenerative strategy using stem cells harvested from the patient’s own bone marrow. These stem cells are injected intrathecally (into the cerebrospinal fluid surrounding the spinal cord) in 20 participants who meet the inclusion criteria for the trial. This is an open label safety and tolerability study and all activities are conducted at Tisch MS Research Center of New York and affiliated International Multiple Sclerosis Management Practice. The interim analysis reports on the first nine patients that have received at least one treatment of stem cells.
Study patient Vicky Gill, a married mother of two whose husband, Michael, also has MS, has already experienced the positive benefits of the therapy. “For the past six years, I would fall frequently, had very limited movement in my legs and always walked with my cane. After just two stem cell treatments, I am now gaining sensation and function I thought was totally lost, have not had any recent falls and at times don’t need a cane at all.”
Trial patients receive three rounds of injections at three month intervals. Safety and efficacy parameters will be evaluated in all patients through regular follow-up visits. Dr. Sadiq plans to continue and complete the Phase I study and if these positive trends continue, move on to a multi-center Phase II efficacy trial.
For more information on this study visit: www.tischms.org
Interim Results of the Tisch MSRCNY stem cell trial presented at the AAN Meeting:
(click on above image to download)
ABOUT TISCH MS RESEARCH CENTER OF NEW YORK
For over twenty years, Saud A. Sadiq, MD, FAAN has believed that combining excellence in clinical care with innovative research targeted at finding the cure for multiple sclerosis would set an exemplary standard in the treatment of MS patients. Today, Tisch MS Research Center of New York embodies this new model of healthcare, with seamless interaction between the practice and the research. Dr. Sadiq and his team of scientists help those with MS by conducting cutting-edge, patient-based research to ensure unparalleled care. The unity of purpose and ease of communication between the non-profit research center and its affiliated clinical practice, the International Multiple Sclerosis Management Practice enables efficient testing of new MS treatments and accelerates the pace at which research discoveries move from lab bench to bedside. Tisch MS Research Center of New York aims to identify the disease trigger so that a cure for multiple sclerosis becomes reality.
###
Support our Research
CLICK HERE TO DONATE TO TISCH MSRCNY
Media Inquiries: Pamela Levin, RN
646-667-3891, plevin@tischms.org
Tisch MS Research Center of New York
521 West 57th Street
4th Floor
New York, NY 10019
(646) 557-3900
Support Tisch MS
Support Tisch MS and our innovative research leading to treatments that improve the lives of patients.